High Prevalence of Atopy in Severe Persistent Pediatric Asthmatics in Memphis, Tennessee
Jennifer Lan, Hoi Sing Chung, Karen Maltby, Charles Stewart, John Vickery, Christie Michael and Betty Lew D
DOI10.21767/2471-304X.100015
Jennifer Lan1, Hoi Sing Chung2, Karen Maltby1, Charles Stewart1,3, John Vickery1, Christie Michael1 and Betty Lew D1*
1Department of Allergy and Immunology, The University of Tennessee Health Science Center, Memphis, TN, USA
2Loewenberg School of Nursing, The University of Memphis, Memphis, TN, USA
3College of Nursing, The University of Tennessee Health Science Center, Memphis, TN, USA
- *Corresponding Author:
- Betty Lew D
MD, Department of Pediatrics
Division of Allergy/Immunology
The University of Tennessee Health Science Center
CFRI, Memphis, TN
United States of America
E-mail: dlew@uthsc.edu
Received date: June 13, 2016; Accepted date: June 25, 2016; Published date: June 30, 2016
Citation: Lan J, Chung HS, Maltby K, et al. High Prevalence of Atopy in Severe Persistent Pediatric Asthmatics in Memphis, Tennessee. Insights Allergy Asthma Bronchitis. 2016, 2:1.
Abstract
Objective: Asthma is a heterogeneous disease that affects millions of adults and children in the United States. The patho-mechanism of asthma is still being elucidated and therapies that control this disease have limitations. It is important to be able to distinguish various asthma phenotypes to allow personalized treatments.
Methods: Pediatric asthmatics from one single institution were enrolled for genomic DNA repository study if they fit the WHO guidelines for severe persistent asthmatics. Various personal health information of the first 100 children were reviewed including gender, age, smoke exposure, lung function test, ICU admissions, controller medication, insurance, body mass index, and allergy skin testing results.
Results: Review of the cohort showed a younger population with a median age of 9 years old. Most importantly, 97% of the patients were found to be atopic. Other findings include a predominantly African American population (89%), mainly holding public insurance (76%), and a significantly overweight and obese (56%) cohort.
Conclusion: The rate of atopy in our cohort of pediatric severe asthmatic children is extremely high, likely due to a warm and humid climate leading to high pollination, mold spores and dust mite content. Early aggressive environmental control and allergen immunotherapy, weight control and social changes are needed. Keywords: Severe asthma; Pediatric; Phenotype; Atopy; Obesity
Keywords
Severe asthma; Pediatric; Phenotype; Atopy; Obesity
Introduction
Asthma is an extremely prevalent disease that affects approximately 24 million adults and children in the United States with significant morbidity and mortality for those who suffer from the disease. According to the Centers for Disease Control and Prevention, asthma is the most common chronic disease responsible for hospitalization in children and affects about 8.6% of the pediatric population [1]. Globally, there are 300 million asthmatics based on a World Health Organization (WHO) [2]. Significant impact on quality of life and health economics cannot be underestimated [1]. Even though multiple genes and environmental factors have been identified, asthma still remains a serious health problem with no curative therapy. Asthma is a complex disease that has multiple components to its patho-mechanism along with many phenotypes and endotypes, making treatment for severe persistent asthma challenging for practitioners.
Those with inherently severe disease are categorized as severe persistent asthmatics. Severe asthmatic patients represent about 5 to 10% of all asthma, but they are responsible for a disproportionate share of the health care costs and morbidity associated with the disease [3-5]. Therefore, studies on severe asthmatics can provide a greater clinical impact compared to those with mild to moderate asthma who respond well to current therapies. These patients have symptoms throughout the day, limitations on daily activity, frequent use of rescue medication, and often difficult to control with standard medication. It is unclear as to why this group is difficult to control, possibly from severe inherent genetic predisposition, environment and social obstacles. Due to the heterogeneity of the disease, multiple routes of evaluation are required to obtain a clearer clinical picture. To date, several methods have been evaluated to determine phenotype, endotype, evaluation of steroid response, evaluation of airflow, evaluation of fungal sensitization, etc [6]. Most recently, clustering approaches were applied to identify more variable clusters [7].
In order to better assess severe asthma in Memphis, TN and the surrounding Mid-South Delta region in US, a pilot study was undertaken to capture a sample of severe persistent asthmatics in this region. The ultimate goal of this study is to evaluate their Whole Exome Sequencing in comparison with those without asthma that will be compared to their clinical phenotype/ endotype in future. Here we present clinical characterization of this cohort highlighting an alarmingly high prevalence of atopy in young children in this southern region of US.
Methods
In our single-center pilot study, 100 patients with severe asthma ranging in ages from 3-18 years old at Le Bonheur Children’s Hospital, the sole free standing children’s hospital in Memphis, TN, were enrolled. The University of Tennessee Institutional Review Board approved this study and written consent was obtained from legal guardian before proceeding. Ninety-seven patients were enrolled from the pediatric asthma clinic, a clinic jointly run by the Pulmonology and Allergy divisions. An additional 3 patients were enrolled when they were admitted to the hospital. To meet criteria, patient had to meet any of the following four criteria, discussed at the WHO Consultation on Severe Asthma. At this meeting, the WHO helped produce some consensus as to the definition of “severe asthma” including those untreated, difficult to treat, and treatment resistant [8]. Patient met criteria for severe asthma if their history includes: an asthma-related ICU admission, be on a minimum daily requirement of inhaled corticosteroids of 800 mcg for greater than 6 months or at least 3 courses of oral steroid bursts per year. Exclusion criteria included any other chronic lung disease, chronic steroid therapy for other diseases, or informed consent was unable to be obtained. Out of 104 patients that met inclusion criteria, four patients declined to participate. For patients enrolled, 5 mL of blood were obtained for later genetic analysis. Phenotypic characteristics were obtained from above patients via electronic health record review at the time of enrollment. Data extracted include gender, age, tobacco smoke exposure, lung function test, ICU admissions, controller medication type, insurance type, body mass index (BMI), and allergy skin testing (SPT, skin puncture test by QUINTEST®, hollisterco.com, Spokane, WA). A wheal size of ≥ 3 mm above the diluent reaction was considered a positive test.
Results
Interestingly, this population is noted to have multiple aeroallergen sensitizations. Out of the 97 patients who had aeroallergen testing, only 3 patients did not have any sensitivity detected on SPT, indicating that 97% of the population had at least one aeroallergen sensitization (Figure 1). Ninety percent of those tested were sensitive to more than one aeroallergen category, with 20% of the population allergic to all categories (Figure 2). The majority of the patients had both perennial (92%) and seasonal allergies (89%) (Figure 1). ICU admissions were prominent with peaks in the spring and the fall, as expected (data not shown).
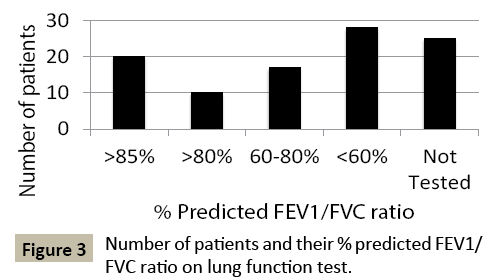
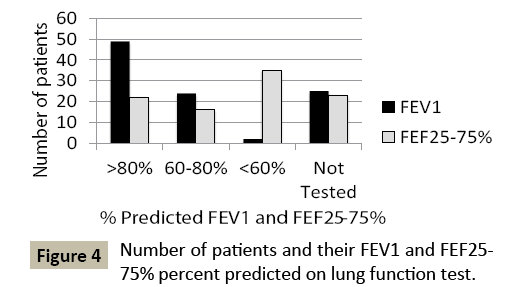
Often in children with asthma, pulmonary function tests (PFTs) are normal. Out of those who were able to perform PFTs, the majority (69%) had FEV1/FVC ratios that categorized them as persistent asthma (FEV1/FVC <85% predicted) (Figure 3). 51% of those enrolled had PFT results that placed them in the severe persistent category (FEV1/FVC <60% predicted). Moreover, 48% patients had significant small airway reduction (FEF25-75% <60% predicted) (Figure 4). Approximately 25% of the patients did not have PFTs at the time due to age or inability to perform.
From a demographics perspective, our asthmatic population is predominantly male at 72% versus 28% female. The population is also predominantly African American, making up 89% of our population (Table 1). The mean age of enrollees is 9.86 years old and median age is 9 years old. Fifty-six percent of the population is categorized as either overweight or obese, with 67% of that population meeting criteria for severe obesity (>99th percentile BMI for age and gender) (Table 1). From a social perspective, the majority of the population enrolled has some form of public insurance, with only 24% owning private insurance (Table 1). A quarter of the asthmatics reports smoke exposure at home per parental history.
| Race N/% | Gender N/% | BMI N/% | Insurance N% |
|---|---|---|---|
| AA89 | M72 | Normal 43 | Pubic 76 |
| C8 | F 28 | Overweight 15 | Private 24 |
| H 2 | Obese6 | ||
| A 1 | Severely Obese 35 | ||
| Unknown1 |
AA: African American; C: Caucasian; H: Hispanic; A: Asian; BMI: Body Mass Index
Table 1: Characteristics of 100 pediatric severe persistent asthmatics in Memphis, TN.
Discussion
Memphis, TN has one of the largest childhood asthma burdens in the state of Tennessee. In 2009, the Tennessee Department of Health reported Shelby County, the county in which Memphis city resides, as one of the top 5 counties with the greatest childhood asthma burden in the state. Besides having one of the highest prevalence of asthma, it also appears to have the highest healthcare utilization secondary to inpatient asthma hospitalization and asthma emergency department (ED) visits in the state [9]. Therefore, it is important to analyze and describe this population to see what may be contributing to this severe burden.
The most interesting observation extracted from this population is the high atopy prevalence. The rate of atopy attributable to symptomatic asthma (regardless severity) is highly variable worldwide (0% in Ankara, Turkey; 70% in the US; 93.8% in Guangzhou China) [10]. Previous asthma phenotype studies have demonstrated that there is a cluster of allergic asthmatics and within that cluster, those with early onset atopic asthma have been frequently reported, that may be consistent with our patient population [11]. Most recently Fitzpatrick has reported a high prevalence of allergen sensitization among severe asthmatics in the NHLBI’s Severe Asthma Research Program (SARP, 2001-2011). Atopy profile of our cohort shows much higher rates of sensitization to all categories of allergens than their cohort [89% versus 76% (indoor allergens), 78% versus 49% (mold), 79% versus 49% (pollens)] [12]. Almost every patient in our cohort had at least one aeroallergen sensitivity, with the majority sensitive to multiple aeroallergens. The majority of the patients had seasonal allergen sensitivities, which correlated with the seasonal variations in the number of ICU admissions. This coincides with previous studies showing children appear to have more hospitalizations in the fall and increased inhaled corticosteroids in the spring and summer [13]. This may indicate that seasonal allergen variation play an important role in asthma exacerbations and severity.
Our group has a large seasonal allergen burden in addition to a large perennial allergen burden, particularly to cockroach and dust mite. Studies have shown that the higher the level of dust mite and cockroach allergen exposure, the more likely the patient is to have a positive allergy skin test response, indicating that our cohort is being exposed to a substantial amount of these aeroallergens, causing them to be sensitizied [14,15].
Regarding a possibility of selection bias towards atopic patients, only three out of the 100 patients in our cohort were obtained upon Allergy/Immunology inpatient consultation. The majority of the patients were selected from the asthma clinic, which is jointly run by both the Pulmonology and Allergy team, therefore, there were non-atopic asthma phenotypes in the selection pool from which these patients were drawn but non-atopic asthmatics were not as severe as the current study cohort. Furthermore, all ICU admissions due to status asthmatics at Le Bonheur Children’s Hospital result in an Allergy/Immunology consult despite asthma phenotype, eliminating any bias towards only atopic asthmatics upon consultation.
The significant atopic nature in our population may help support the hypothesis that those with allergy driven disease in children in general are more inherently severe and therefore difficult to treat. However, the consensus on the above statement is still unsettled. A recent review on allergic asthma phenotype actually showed the opposite, demonstrating that allergic asthmatics are less severe [11]. Another study showed those with severe asthma tends to have greater magnitude of atopy and lower lung function than those with mild moderate asthma [4]. Other studies have shown that atopy with high IgE levels, eosinophilia, or skin prick positivity is associated with increased risk of persistent wheeze [16]. It is likely that the timing of study in the course of disease progression may influence the outcome of the study. Obviously, further studies are needed in young children to help elucidate the role of atopy in the development and progression of severe asthmatics.
Along with the high prevalence of atopy in this cohort, one must also take into account the young age of our cohort. The range of enrollees spanned a wide age range from 3 to 18 years old. However, both our mean and median age is around 9 year of age, which may give an insight as to the pathogenesis of asthma in this cohort. Studies have found that the first few years of life presents a window of vulnerability during which airway injury can result in persistent airway dysfunction [17,18]. Lung function growth patterns are usually established as early as birth, but certainly within the first 6 years of life [19]. Those with earlyonset wheeze along with atopy have been shown to have lung function abnormalities by early adolescence [16]. Those with chronic wheeze as young as one to three year of age already have reticular basement membrane thickening and eosinophilic inflammation [19]. Another study has shown that preschool children with chronic wheeze and atopy have larger area of airway smooth muscle, predisposing themselves to higher bronchial hyperresposniveness than those with nonatopic asthma [20]. Because atopy develops during this window of vulnerability, along with possible microbial illnesses, we must seek early intervention to reverse the course.
There is also a second period of vulnerability around adolescence that may also increase development of persistent wheeze into adulthood [21]. In this second window, allergic sensitizations along with weight gain and smoking have been found to attribute to lung injury during this adolescent period of lung vulnerability [16]. These above finding implicate important clinical interventions for our young and atopic patient population. First, the young age of our cohort suggests that early allergen sensitization is the primary contributing factor to their severe symptoms. With that in mind, early intervention in their first few years of life may be essential in preventing disease or halting it from progressing. Such interventions as environmental control and allergen immunotherapy during preschool years may modify the progression of their asthma in our patient population.
Another way to help characterize the innate severity of asthma of our cohort is by looking at abnormalities of their PFT. The 2007 National Heart Lung and Blood Institute Guidelines for the Diagnosis and Treatment of Asthma Expert Panel Report published cut offs for various degrees of severity based on FEV1 and FEV1/ FVC ratios [22]. Unfortunately, many times in asymptomatic children, spot check FEV1 is often normal even in children with uncontrolled symptoms. Even though no recommendation exists as to the utility of the percent predicted of the lower airways measured on PFT (FEF25-75%), there have been some studies showing that children with reduced lower airway patency have worse clinical outcomes, requiring more frequent hospitalization, steroid use, and ED visits [23]. Almost half of our patients who were able to perform PFT showed significantly reduced small airway flow, despite normal FEV1, indicating that they are still at risk of significantly worse clinical outcomes. Solely using PFT standards and cut offs such as FEV1 and FEV1/FVC ratio as a way to classify patients can miss many severe asthmatics in children. Like with many chronic conditions, it is important that physicians use multiple age-appropriate tools to assess severity of disease and control such as with a combination symptom report, PFT, diurnal variation of peak flow rate, airway hyper-responsiveness and inflammatory markers such as eosinophils, and fractional exhaled nitric oxide [24].
The fact that there is a high prevalence of obesity in our cohort adds to comorbidities; 56% of the group is classified as either overweight or obese, the majority being severely obese. Interestingly, a study comparing overweight asthmatic children to lean asthmatic children, found that overweight asthmatics reported more albuterol use compared to their leaner counterparts despite having similar lung function results, reduced methacholine responsiveness, and lower fractional excretion of nitric oxide results. The overweight children may display poorer asthma control due to exertional symptoms and/or gastroesophageal reflux [25]. Moreover, obesity may be contributing to asthma from multiple patho-mechanisms including overall mechanical effects on lungs, chronic inflammation, and increased airway hypersensitivity [26]. Obesity certainly is a compounding factor in the difficulty of controlling highly atopic severe asthmatic children.
Our demographics show that severe asthma is more prevalent among boys than girls, consistent with the report by Guilbert et al. [27] and among African Americans compared to Hispanic and Caucasian children. The proportion of African Americans in our cohort is even higher than the average African American population here in Memphis, TN. According to the 2010 US Census data for Memphis, TN, 63.3% of the population is African American [28]. Our cohort is 26% percent higher than the TN statistics, signifying that the African American asthmatics in Memphis, TN are more prone to severe asthma. Racial disparity in asthma is commonly recognized (9.9% vs. 7.6% vs. 6.7%, Black vs. White vs. Hispanics, respectively), yet there are only a few studies on racial disparity in childhood asthma and atopy despite CDC statistics between 2012-2014 (4 times higher hospital admission rate and 10 times higher mortality rate compared to non-Hispanic White children) [1]. A study by Wegienka et al. on birth cohort of young children (age 2 yrs) found atopy prevalence rate among African American children twice as high as compared to White children in Detroit, MI (21.7% versus 11.0%) [29]. In a childhood asthma cohort with recurrent wheezing Guilbert et al. [27], atopy rate was 60.7% although their cohort was predominantly White with only 11.9% non-hispanic Black. Our cohort is unique in that children with severe asthma were targeted and a remarkably high atopy rate was found in a predominantly African American cohort. Moreover, the majority of our patients have public insurance. This correlates with previous studies that those with public insurance have a higher rate of asthma compared to those with private insurance (13.6% vs. 7% in TN) [9]. Memphis itself, compared to the rest of TN, has a larger poverty population with 26.9% of the population living below poverty compared to 17.6% in the rest of TN [28]. Another recent study has reported that living in the inner city does not necessarily increase the risk of asthma, but being African American, Puerto Rican, or having lower household income does, regardless of residence (inner city versus non-inner city) [30]. Undoubtedly, poverty is an additional impediment to environmental control and therapeutic intervention.
It is well known that smoke exposure contributes to the development of asthma. Burke et al. meta-analysis showed that even passive exposure to smoke (both pre- and post-natal) increases asthma development [31]. Furthermore, morbidity and mortality increases for patients with asthma who smoke or are around smokers [32,33]. Interestingly, only 25% of our patients reported a known smoke exposure. However, it is possible that this is an underestimate of actual smoke exposure since most studies have shown underreporting of second hand smoke [34].
Conclusion
Severe asthma is a multifactorial disease, making it difficult to treat effectively by protocol or guidelines. In this population in Memphis, TN and the surrounding Mid-South Delta region, there appear to be many elements that we can address as providers, such as obesity, smoke exposure, access to healthcare, and poverty. However, the most important takeaway points from this cohort is that atopy and allergen exposure early in life most likely has a large impact on asthma severity in this patient population.
The findings described here indicate that there are multiple personalized venues for intervention including aggressive early environmental changes and allergen immunotherapy during preschool years, weight control and social changes. Expeditious FDA approval of biologics for young children along with application of genomic information would further propel severe asthma management toward precision medicine.
References
- CDC (2013) National Health Interview Survey data: Asthma: Data, Statistics, and Surveillance. Atlanta, GA: US Department of Health and Human Services. Available at https://www.cdc.gov/asthma/asthmadata.htm. Accessed on July 14, 2016.
- WHO (2007) Global surveillance, prevention and control of chronic respiratory diseases: a comprehensive approach.
- Moore WC, Peters SP (2006) Severe asthma: An overview. J Allergy Clin Immunol 117: 487-494.
- Cowan K, Guilbert TW (2012) Pediatric asthma phenotypes. Curr Opin Pediatr 24: 344-351.
- Kupczyk M, Wenzel S (2012) US and European severe asthma cohorts: what can they teach us about severe asthma? J Intern Med 272: 121-132.
- Bush A, Fleming L (2011) Phenotypes of refractory/severe asthma. Paediatr Respir Rev 12: 177-181.
- Wu W, Bleecker E, Moore W, Busse WW, Castro M, et al. (2014) Unsupervised phenotyping of severe asthma research program participants using expanded lung data. J Allergy Clin Immunol 133: 1280-1288.
- Bousquet J, Mantzournais E, Cruz AA, Ait-Khaled N, Baena-Cagnani CE, et al. (2010) Uniform definition of asthma severity, control, and exacerbations: Document presented for the World Health Organization Consultation on Severe Asthma. J Allergy Clin Immunol 126: 926-938.
- Audrey MB (2009) Office of Policy, Planning and Assessment. Childhood Asthma in Tennessee 1997-2007. Tennessee Department of Health, Nashville, TN.
- Weinmayr G, Forastiere F, Weiland SK, Rzehak P, Abramidze T, et al. (2008) International variation in prevalence of rhinitis and its relationship with sensitization to perennial and seasonal allergens. Eur Respir J 32: 1250-1261.
- Schatz M, Rosenwasser L (2014) The allergic asthma phenotype. J Allergy Clin Immunol Pract 2: 645-648.
- Fitzpatrick AM (2016) Severe Asthma in Children: Lessons Learned and Future Directions. J Allergy Clin Immunol Pract 4: 9-11.
- Teach SJ, Gergen PJ, Szeifer SJ, Mitchell HE, Calatronia A, et al. (2015) Season risk factors for asthma exacerbations among inner-city children. J Allergy Clin Immuno 135: 1465-1473.
- Huss K, Adkinson NF Jr, Eggleston PA, Dawson C, Van Natta ML, et al. (2001) House dust mite and cockroach exposure are strong risk factors for positive allergy skin test responses in the Childhood Asthma Management Program. J Allergy Clin Immunol 107: 48-54.
- Gruchalla RS, Pongracic J, Plaut M, Evans R, Visness CM, et al. (2005) Inner City Asthma Study: Relationships among sensitivity, allergen, exposure, and asthma morbidity. J Allergy Clin Immunol 115: 478-485.
- Grad R, Morgan WJ (2012) Long-term outcomes of early-onset wheeze and asthma. J Allergy Clin Immunol 130: 299-307.
- Martinez FD (2011) New insights into the natural history of asthma: primary prevention on the horizon. J Allergy Clin Immunol 128: 939-945.
- Martinez FD, Stern DA, Wright AL, Taussig LM, Halonen M (1998) Differential immune responses to acute lower respiratory illness in early life and subsequent development of persistent wheezing and asthma. J Allergy Clin Immunol 102: 915-920.
- Saglani S, Payne DN, Zhu J, Wang Z, Nicholson AG, et al. (2007) Early detection of airway wall remodeling and eosinophilic inflammation in preschool wheezers. Am J Respir Crit Care Med 176: 858-864.
- Lezmi G, Gosset P, Deschildre A, Abou-Taam R, Mahut B, et al. (2015) Airway Remodeling in Preschool Children with Severe Recurrent Wheeze. Am J Respir Crit Care Med 192: 164-171.
- Strunk RC, Weiss ST, Yates KP, Tonascia J, Zeiger RS, et al. (2006) Mild to moderate asthma affects lung growth in children and adolescents. J Allergy Clin Immunol 118: 1040-1047.
- National Asthma Education and Prevention Program (2007) Expert Panel Report 3 (ERP-3): Guidelines for the diagnosis and management of asthma- summary report 2007. J Allergy Clin Immunol 120: 94-138.
- Rao DR, Gaffin JM, Baxi SN, Sheehan WJ, Hoffman EB, et al. (2012) The utility of forced expiratory flow between 25% and 75% of vital capacity in predicting childhood asthma morbidity and severity. J Asthma 49: 586-592.
- Nuijsink M, De Jongste JC, Pijnenburg MW (2013) Will symptom-based therapy be effective for treating asthma in children? Curr Allergy Asthma Rep 13: 421-426.
- Lang JE, Hossain MJ, Lima JJ (2015) Overweight children report qualitatively distinct asthma symptoms: Analysis of validated symptom measures. J Allergy Clin Immunol 135: 886-893.
- Permaul P, Kanchongkittiphon W, Phipatanakul W (2014) Childhood asthma and obesity- what is the true link? Ann Allergy Asthma Immunol 113: 244-246.
- Guilbert TW, Morgan WJ, Zeiger RS, Bacharier LB, Boehmer SJ, et al. (2004) J Allergy Clin Immunol 114: 1282-1287.
- United States Census Bureau. State and County Quick Facts (2010) Available at: https://quickfacts.census.gov/qfd/states/47/4748000.html. Accessed June 21, 2015.
- Wegienka G, Havstad S, Joseph CLM, Zoratti E, Ownby D, et al. (2012) Clin Exp Allergy 42: 909-917.
- Keet CA, McCormack MC, Pollack CE, Peng RD, McGowan E, et al. (2015) Neighorhood poverty, urban residence, race/ethnicity, and asthma: Rethinking the inner-city asthma epidemic. J Allergy Clin Immunol 135: 655-662.
- Burke H, Leonardi-Bee J, Hashim A, Pine-Abata H, Chen Y, et al. (2012) Prenatal and passive smoke exposure and incidence of asthma and wheeze: systematic review and meta-analysis. Pediatrics 126: 735-744.
- Polosa R, Knoke JD, Russo C, Piccillo G, Caponnetto P, et al. (2008) Cigarette smoking is associated with a greater risk of incidient asthma in allergic rhinitis. J Allergy Clin Immunol 121: 1424-1428.
- Eisner MD (2008) Passive smoking and adult asthma. Immunol Allergy Clin North Am 28: 521-537.
- Boyaci H, Etiler N, Duman C, Basyigit I, Pala A (2006) Environmetnal tobacco smoke exposure in school children: Parent report and urine cotinine measures. Pediatr Int 48: 328-382.
Open Access Journals
- Aquaculture & Veterinary Science
- Chemistry & Chemical Sciences
- Clinical Sciences
- Engineering
- General Science
- Genetics & Molecular Biology
- Health Care & Nursing
- Immunology & Microbiology
- Materials Science
- Mathematics & Physics
- Medical Sciences
- Neurology & Psychiatry
- Oncology & Cancer Science
- Pharmaceutical Sciences