Peritoneal Mast Cell Stabilization Potential of Ziziphus Xylopyrus (Retz) Wild Extract in Rat Mesenteric Model
Saurabh Gupta,K. Rama Satyanarayana Raju,Shashank Mulukutla, Nilesh Ambhore,Yamini Parmar,Swati Khare,Vidhi Sharma,Richa Pathak,Paramjeet Devda and Tagis Mehar
DOI10.21767/2471-304X.100007
Saurabh Gupta1*, K. Rama Satyanarayana Raju2, Shashank Mulukutla2, Nilesh Ambhore2, Yamini Parmar1, Swati Khare1, Vidhi Sharma1, Richa Pathak1, Paramjeet Devda1and Tagis Mehar1
1Department of Pharmacology, Indore Institute of Pharmacy, Pithampur road, Opp. IIM, Rau, Indore, M.P, India
2Department of Pharmacology, JSS College of Pharmacy, Off Campus JSS University, Ooty, Tamil Nadu, India.
- *Corresponding Author:
- Saurabh Gupta
Associate Professor and Head, Department of Pharmacology
Indore Institute of Pharmacy, Pithampur Road
Opp. IIM, Rau, Indore (M.P.) 453331, India
Tel: 0091-9407178028
E-mail: saurabhgupta80@gmail.com
Received: September 14, 2015, Accepted: October 19, 2015, Published: October 21, 2015
Citation: Gupta S, Raju KRS, Mulukutla S, et al. Peritoneal Mast Cell Stabilization Potential of Ziziphus Xylopyrus (Retz) Wild Extract in Rat Mesenteric Model.Insights Allergy Asthma Bronchitis. 2015, 1:1. doi:10.21767/2471-304X.100007
Abstract
Objective: To investigate the peritoneal mast cell stabilization activity of Ziziphus xylopyrus extracts.
Materials and Methods: Ziziphus Xylopyrus L. (family- Rhamnaceae) stem part was successively extracted with ethanol and aqueous to prepare extract of the plant. The extracts of Ziziphus Xylopyrus (Z. xylopyrus) were evaluated for stabilization of mast cell in rat for the evaluation of Hypersensitivity type I allergic reaction in animal models. The extract of Z. xylopyrus ethanolic and aqueous (1, 10 and 100 μg/ml) was evaluated for peritoneal mast cell stabilization activity in rat mesenteric preparation induced by C 48/80.
Result: Preliminary phytochemical analysis revealed the presence of carbohydrates, fixed oil, proteins, alkaloids, glycosides, flavonoids and phenolic compounds. The ethanolic and aqueous extracts of Z. xylopyrus showed dose dependent increase in the number of intact cells when compare with C 48/80 at the concentration of 10 and 100 μg/ml. It virtues further work towards the isolation of phyto constituents from this plant.
Conclusion: This finding provides evidence that the Z. xylopyrus inhibits mast cell-derived immediate-type allergic reactions and mast cell degranulation. Z. xylopyrus has a potential as allergic anti-asthmatic agent.
Keywords
Compound 48/80, Ethanolic extract, Mast cell, Z. xylopyrus
Introduction
Ziziphus xylopyrus (Retz) Willd (family- Rhamnaceae) is a large, straggling shrub or a small tree, armed with spines, up to 4 m in height. Z. xylopyrus is commonly known as Jujab [1]. It is found in Pakistan and China, North-Western India, Utter Pradesh, Bihar, Central and South India [2]. Z. xylopyrus is used in Pyorrhoea and to check oogenesis [3,4]. The bark is used for its astringent activity and as dental sticks for teeth cleaning [5,6]. India traditionally fresh stem bark of Z. xylopyrus species are chewed with 1-2 peppers (Piper nigrum) for the treatment of cough. Bark and leaf powder paste are applied externally for chest pain arising from cough [7]. This plant is widely used in Turkish medicine as a potent sedative [8].
The previous literature reported that Z. xylopyrus leaves contain quercetin and quecitrin. The bark contains tannins (7.2%) 7, 3, 4-trihydroxy fl avan-3, 4-diol and oleanic acid [9]. The plant contain Xylopyrine-A and Xylopyrine-B, the two new 13-membered cyclopeptide alkaloids are also present in Ziziphus species i.e. Z. xylopyra [10]. The phytochemical screening of Z. xylopyrus leaves extract with various solvents showed the presence of alkaloids, carbohydrates, steroids and sterol, glycosides, saponins, flavonoids, phenolic compounds, triterpenoids [11-13].
Mast cells are significantly found in mucosal tissues at homeostasis and their elevated numbers due to the allergic disorders like asthmatic lungs, inflammatory bowel disease (IBD) of gastrointestinal tract (GIT) and recto colon cancer. In 1878, scientist Ehrlich in his doctoral thesis describes first about mast cells and its features. He observe mast cell having unique staining characteristics and large granules, they termed mast cell as “Mastzellen” which means well-fed cells, because their cytoplasm filled with granular material. Now a day mast cells are also considered to be part of the immune system because its participated in host defense mechanism [14]. Mast cells play a povitel role in initiating inflammation and immediate type I hypersensitivity allergic reactions [15]. Mast cells initiate the allergic reaction due to that release of following inflammatory mediators such as, histamine, proteases, chemotactic factors, cytokines like (Interleukins -6 (IL-6), IL-13, IL-4 etc.) and metabolites of arachidonic acid. Different authors reported that histamine release when the body encounters a toxic substance even it’s also released when mast cells detect injury [16]. The present study was proposed to find out the alternative herbal medicines which counter the allergic response by reducing the side effect of synthetic drugs. In this direction we evaluate the mast cell stabilization property of Z. xylopyrus stem extracts on peritoneal mesentery preparation.
Materials and Methods
Collection
Fresh stem parts of Ziziphus xylopyrus (Retz) Willd stem were collected in the month of July, 2014, from Tirupati district, Andhra Pradesh, India. Dr. K. Madhava Chetty, Associate professor and Head, Department of Botany, Sri Venkateswara University, Tirupati, India authenticated the collected plant. Voucher specimen has been preserved in our laboratory (SVU/ SC/30/98/13-14) for future reference.
Chemicals
Ethanol, Sodium bi Carbonate, Potassium chloride, Sodium Chloride, Calcium chloride, Dextrose, Xylene, Acetone, Molish reagent, Millions reagent and Anesthetic ether etc. were purchased from E-Merck (India) Ltd., Mumbai, India. O-toludine blue and Compound 40/80 were purchased from sigma Pvt. Ltd. etc. All chemicals used were of analytical grade. Standard drug Disodium cromogylate (DSCG) was obtained from Cipla Pvt. Ltd. as a gift sample.
Animals
Healthy male albino Wistar strain rats of (170–200 g) were obtained from the animal house, J.S.S. College of Pharmacy, Ootacamund, India, and were maintained under standard environmental conditions (SEC) (22–28 °C, relative humidity 60- 70%, 12-hr dark: 12-hr light cycle) and were fed with standard rat feed (SRD) ad libitum (M/S Hindustan Lever Ltd., Bangalore, India) and water. The experiments were conducted as per the guidelines of CPCSEA, Chennai, India (Approval no. JSSCP/IAEC/ Ph.D/P.Cog/02/2011-12).
Preparation of extracts
The Z. xylopyrus stem part was cleaned thoroughly with tab water to remove unwanted matter, dried the whole material in shade, grounded the material into coarse powder with mechanical grinder and passed through sieve no. 40. Further, plant material was extracted with cold maceration process using absolute ethanol (99.98% v/v) by intermittent shaking for 10 days, filtered and dried marcleft. Again the resultant residue was macerated The Z. xylopyrus stem part was cleaned thoroughly with tab water to remove unwanted matter, dried the whole material in shade, grounded the material into coarse powder with mechanical grinder and passed through sieve no. 40. Further, plant material was extracted with cold maceration process using absolute ethanol (99.98% v/v) by intermittent shaking for 10 days, filtered and dried marcleft. Again the resultant residue was macerated
Qualitative phytochemical screening
Z. xylopyrus extracts were subjected for phytochemical tests to find the presence of primary and secondary matabolite phytochemical constituents according to standard methods [17].
Mast cell stabilization activity
Overnight fasted male Wister rats were sacrificed with excess dose of anesthetic ether under anesthetic chamber. The abdomen was cut open to expose the intestine portion. Pieces of mesentery with connecting lobes of fat and blood vessels were rapidly dissected out and then small pieces of the mesentery were cut and placed in a beaker containing Ringer Locke (in mM: Sodium Chloride 154, Potassium chloride 5.6, Calcium chloride 2.2, Sodium bi Carbonate 6.0 and dextrose 5.5) solution for 30 ± 1 min containing different concentration of Z. xylopyrus plant extracts. Later, the tissues were exposed to compound 48/80 (to promote mast cell degranulation). The tissue was incubated for further 30 ± 1 minute. The pieces of mesentery were removed and placed in a clean slide and trimmed to remove fatty layers. The trimmed tissue was dipped in 4% formaldehyde solution containing 0.1% O-toludine blue for 20-30 min and then the tissue was transferred through acetone and Xylene two times (2 changes each) for 5 ± 1 min. Six pieces of mesentery were used for each concentration of the test substance.
The stained mesentery pieces were focused through a digital light microscope (M/s. Motic, Japan) at 100 x magnification. 100 mast cells were counted, starting from the left hand side of the field and then proceeding clockwise. The number of intact and fragmented or disrupted mast cells was noted. A mast cell was considered disrupted if 4 or 5 granules were found around the mast cells. The percentage of mast cell fragmented or disrupted and of intact mast cells was calculated [18,19].
The experimental groups for were as follows:
The following were the groups used for the study
Group I : Tissues exposed in ringer locke solution only (Vehicle control)
Group II : Tissues exposed to 0.8 μg/ml of C 48/80 only (Negative control)
Group III : Tissues exposed to Disodium cromogylate-DSCG, 1 mg/ ml (Positive control)
Group IV : Tissues exposed in ZXE 1 μg/ml
Group V : Tissues exposed in ZXE 10 μg/ml
Group VI : Tissues exposed in ZXE 100 μg/ml
Group VII : Tissues exposed in ZXA 1 μg/ml
Group VIII : Tissues exposed in ZXA 10 μg/ml
Group VIIII : Tissues exposed in ZXA 100 μg/ml
All the groups except group I and II were later exposed to 0.8 μg/ ml of C 48/80.
Statistical analysis
Statistical analysis was done by using one-way Analysis of Variance (ANOVA) followed by Turkey’s multiple comparison tests. P value<0.05 was considered to be statistically significant. The analysis was carried out using Graph Pad Prism software V.5.04.
Results
Extract recovery percent
The maximum yield was obtained in ZXA extract when compare to ZXE. The extractive values were found to be: 1.84 and 3.37% w/w for ethanol and aqueous respectively.
Qualitative phytochemical screening
The phytochemical screening on Z. xylopyrus revealed the presence of primary metabolites secondary metabolites. Results are mention in (Table 1).
| Phytochemical tests | Ziziphusxylopyrus | |
|---|---|---|
| Stem extract | ||
| ZXE | ZXA | |
| Alkaloids | ++ | + |
| Carbohydrates | + | + |
| Steroids and sterols | + | - |
| Glycosides | - | - |
| Saponins | + | ++ |
| Protein and amino acids | - | - |
| Flavonoids | ++ | + |
| Phenolic | +++ | + |
| Acidic | - | - |
| Fixed oils | + | - |
| Triterpenoids | + | - |
(+): Presence of chemical compounds, (-): absence of chemical compounds.
(+) < (++) < (+++): Based on the intensity of characteristic colour.
Table 1: Qualitative phytochemical analysis of Ziziphus xylopyrus.
Mast cell stabilization activity
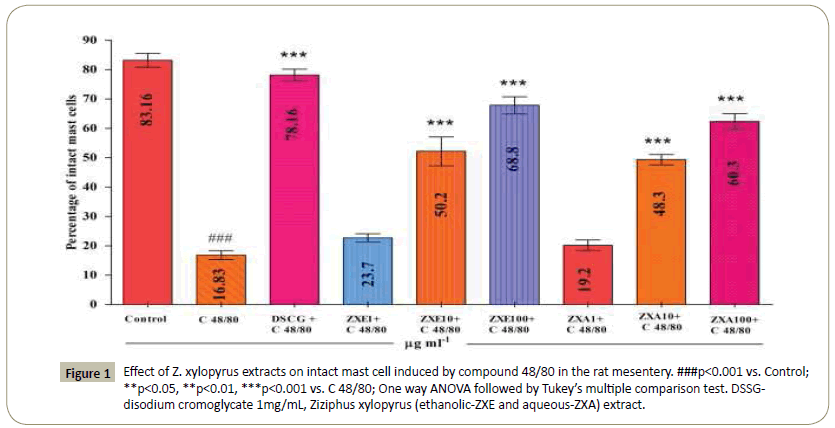
C 48/80, a mast cell degranulating agent, showed significant (p<0.001) increase in degranulation of rat mesentery mast cell, (83.2 ± 1.5), when compared to vehicle control (16.8 ± 2.4). Disodium Chromoglycate (DSCG), a known mast cell stabilizing agent, showed significant (p<0.001) percentage reduction in degranulated mast cells (22.2 ± 1.9), when compared to C 48/80 group (Table 2). At the concentration of 10 and 100 mg/ml, ethanolic, and aqueous extracts of ZXE produced dose dependent and showed significant (P < 0.001) increase in the number of intact cells when compared with C48/80 treated tissue (Figure 1). Among the different extracts ZXE 10 mg/ml (50.2 ± 4.8) and 100 mg/ml (68.8 ± 2.7) showed significant protection of cell as compared ZXA.
Figure 1: Effect of Z. xylopyrus extracts on intact mast cell induced by compound 48/80 in the rat mesentery. ###p<0.001 vs. Control; **p<0.05, **p<0.01, ***p<0.001 vs. C 48/80; One way ANOVA followed by Tukey’s multiple comparison test. DSSGdisodium cromoglycate 1mg/mL, Ziziphus xylopyrus (ethanolic-ZXE and aqueous-ZXA) extract.
| Group | Treatment (N=6) | Concentration µg ml-1 | Percentage of | |
|---|---|---|---|---|
| Intact Mast Cells | Degranulated Mast Cells | |||
| 1 | Vehicle control | - | 83.2 ± 2.4 | 16.8 ± 2.4 |
| 2 | C 48/80 | - | 16.8 ± 1.5### | 83.2 ± 1.5### |
| 3 | DSCG | 1mg ml-1 | 77.8 ± 1.9*** | 22.2 ± 1.9*** |
| 4 | ZXE | 1 | 23.7 ± 1.4 | 76.3 ± 1.4 |
| 5 | ZXE | 10 | 50.2 ± 4.8*** | 49.8 ± 4.9*** |
| 6 | ZXE | 100 | 68.8 ± 2.7*** | 31.2 ± 2.9*** |
| 7 | ZXA | 1 | 19.2 ± 1.7 | 80.8 ± 1.8 |
| 8 | ZXA | 10 | 48.3 ± 1.7*** | 52.7 ± 1.8*** |
| 9 | ZXA | 100 | 60.3 ± 2.7*** | 39.7 ± 2.7*** |
Values expressed as mean ± SEM N=number of rat mesentery per group Statistical significance; ###p<0.001 vs. vehicle control; *p<0.05, ***p<0.001 vs. C 48/80: One way ANOVA followed by Tukey’s multiple comparison test.
DSCG-disodium cromoglycate
Table 2: Effect of Ziziphus xylopyrus extracts on mast cell degranulation induced by C48/80 in the rat mesentery.
Discussion
One of the common diseases that affect human races with diverse manifestations is allergic asthma, which is responsible for significant morbidity with severe economic impact. Various epidemiological studies have identified the cause for an increase in the prevalence of allergic disorders [20]. Some of the postulated reasons are increasing environmental pollution and increased predisposition of individuals producing excessive IgE through a major change in the gene pool and changing life style [21]. Continuous uses of allopathic drugs, which are available in market, develop drug resistance and severe adverse effects which include desensitization of receptor. So there is a need for find alternative herbal medicine which is effective and obtained from natural source to treat allergic asthma. The current therapeutic approaches for the treatment have limitation in their ability to target all the features of the disease. Thus, there is an unmet need for developing new drugs to target these features along with improved efficacy and without side effect.
Due to these reasons, there is a need of new-targeted therapeutic agent required to treat asthma. In this direction now a days potential leads from medicinal plants that may be effective against type I hypersensitivity reactions involving various phases of allergic disorder. On the basis of ethnobotanical claims Z. xylopyrus having antiallergic disorder properties. In the present study Z. xylopyrus extracts were investigated for their in vitro and in vivo antiallergic activity. Efforts were also made to identify the lead phytoconstituents that are responsible for the inhibition of allergic reaction.
The preliminary physicochemical evaluation of the drug is an important parameter in detecting the adulterants or improper handling of drugs and identification of plant material. The results reveal that the ash and extractive values of Z. xylopyrus were found to be within the standard range [22]. The preliminary phytochemical investigations carried out on the extracts of Z. xylopyrus has revealed the presence of carbohydrates, alkaloids, glycosides, saponins, sterols, flavonoids, phenolic compounds and tannins. The phytochemicals tests help in laying down the pharmacopoeial standards [23,24].
Various author reported that from therapeutic point of view, alkaloids, phenolic and flavinods containing plants are used in traditional medicine to treat allergic disorder Gardenia latifolia, Ginkgolides from Ginko biloba leaves have been used to treat asthma. The plants Zizipbus polygala, Panax ginseng and Glycyrrhiza glabra have been useful as antifatigue agents, antiphlogistics, expectorants for bronchitis and asthma, and hepatoprotective etc. [25]. These herbs have already shown interesting results in various target specific biological activities like broncho dilation, mast cell stabilization, anti-anaphylactic, antiinflammatory, antispasmodic, anti-allergic, immunomodulatory and inhibition of mediator’s viz., leukotrienes, lipoxygenase, phosphodiesterase and cytokine, in the treatment of allergic as well as different disordors [26].
Type-1 or Immediate Hypersensitivity Type reaction or Anaphylactic Hypersensitivity reaction consists of various allergic disorders ranging from a serious anaphylactic reaction to an acute pruritis and to chronic condition like allergic asthma. The basic component involved in Type I reaction is mast cell and IgE antibody [15]. In allergic asthma, where type I hypersensitivity reaction occurs, mast cells play a prominent role [27]. Mast cell bind with allergens leads to the generation of a variety of autacoids including histamine, cysteinyl-leukotrienes, prostaglandin-D, cytokines and enzymes such as tryptase etc. During an allergic response, B cells released high affinity IgE receptors (Fc?RI) at the Fc portion, present on the surfaces of the mast cells. Due to the sensitization of the mast cells, which lead the production of histamine, prostaglandins and cytokines etc. When this has happen, the allergen binds to the Fab portion of IgE receptor thereby activating the G protein pathway. So there is increased in Ca2+ inflow. This increased calcium concentration culminates in the degranulation of the mast cell, releasing its constituents [13]. Previous studies indicated that compound 48/80 directly activated G-proteins receptors. It increases the permeability of the lipid bilayer membrane by causing the perturbation. This intracellular calcium is crucial to the degranulation of mast cells [28]. The present study based on the traditional use of Z. xylopyrus in allergic disorder. It was investigating the potential of the extracts in Type I hypersensitivity reaction in animal models. The predictive model for screening mast cell stabilization activity of test agents. [19] In the present investigation, the authors have tried to assess the in vitro mast cell stabilization studies of the extracts ZXE and ZXA observed a significant dose dependant reduction (p<0.001) in mast cell degranulation when compared to C48/80. ZXE extracts (68.8 ± 2.7) demonstrated dose dependent highest mast cell stabilization ability in rat mesentery preparation. The preliminary phytochemical studies reports that the plant contain high amount of flavonoids and phenolic compounds. On the basis of previous reports flavonoids and phenolic compound have been shown to possess smooth muscle relaxant and bronchodilator activity. Flavonoids also inhibit the histamine release induced by C48/80 [29]. The phytochemical screening on Z. xylopyrus revealed the presence of alkaloids, triterpenoids, flavonoids and phenolic compounds [30]. The mast cells play a major role in Type I hypersensitivity-mediated diseases like allergic reaction which leads to cause asthma and rhinitis disorder.
Conclusion
This finding provides evidence that the Z. xylopyrus inhibits mast cell derived immediate type I allergic reactions and mast cell degranulation. Z. xylopyrus will be the potential candidate for allergic and anti-asthmatic activity. Studies are underway to evaluate the efficacy of Z. xylopyrus due to its mast stabilization property in these animal allergic models. It virtues further work towards the isolation of phytoconstituents from this plant.
Acknowledgment
Authors are grateful to JSS College of Pharmacy and JSS University, Mysore for kind support. The authors are also thankful to Cipla Pvt. Ltd. for providing Disodium Cromoglycate standard as a gift to carry out the study. I personally acknowledge to my supervisor Late Dr. M. N. Sathish Kumar for his valuable inputs and guidance for this research work.
References
- Department of AYUSH (2006) Ayurvedic Pharmacopoeia of India. Department of ISM and H, Controller of Publications 5: 47.
- Anonymous (1976) The Wealth of India: Raw Materials Series. Vol. 11 (X-Z). New Delhi: NISCAIR, CSIR pp. 123-4.
- Jain A, Katewa SS, Chaudhary BL, Galav P (2004) Folk herbal medicines used in birth control and sexual diseases by tribals of southern Rajasthan, India. J Ethnopharmacol 90:171-177.
- Meena AK, Rao MM (2010) Folk herbal medicines used by the Meena community in Rajasthan. Asian J Tradit Med 5:19-31.
- Dash SK, Padhy S (2006) Review on ethnomedicines for diarrhoea diseases from Orissa prevalence versus culture. J Hum Ecol 20:59-64.
- Naidu KA, Khasim SM (2010) Contribution to the floristic diversity and ethno botany of Eastern Ghats in Andhra Pradesh, India. Ethnobotanical Leaflets 14:920-941.
- Reddy KN, Reddy CS, Trimurthulu G (2006) Ethno botanical survey on respiratory disorders in Eastern Ghats of Andhra Pradesh, India. EthnobotanicalLeaflets 10:139-148.
- Jagtap SD, Deokule SS, Bhosle SV (2006) Some unique ethnomedicinal uses of plants used by the Korku tribe of Amravati district of Maharashtra, India. J Ethnopharmacol 107:463-469.
- Yadav M, Meena AK, Rao MM, Kapil P, Panda P, et al. (2011) Review on Ziziphusxylopyrus: A potential traditional drug. J Pharm Res 4:922-923.
- Singh AK, Pandey MB, Singh VP, Pandey VB (2007) Xylopyrine-A and xylopyrine-B, two new peptide alkaloids from Zizyphusxylopyra. Nat Prod Res 21:1114-1120.
- Jain S, Sharma C, Khatri P, Jain A, Vaidya A (2011) Pharmacognostic and Phytochemical Investigations of the leaves of Ziziphusxylopyrus (Retz ) Wild. Int J Pharm PharmSci 3:122-125.
- Gandagule UB, Duraiswamy B, Zalke AS, Qureshi MA (2013) Pharmacognostical and phytochemical evaluation of the leaves of Ziziphusxylopyrus(Retz) Willd. Ancient Sci Life 32:245-149.
- Sharma VK, Chauhan NS, Lodhi S, Sighai AK (2009) Anti-Depressant Activity of Ziziphusxylopyrus. Int J Phytomedicine 1:12-17
- Kawa A (2012) The role of mast cells in allergic inflammation. Respir Med 106:9-14.
- Peachell P (2005) Targeting the mast cell in asthma. CurrOpinPharmacol5:251-256.
- Skidgel RA, Erdos EG (2006) Histamine, bradykinin and their antagonists In: Brunton LL, Lazo JS, Parker KL, editors. Goodman and Gilman’s: The Pharmacological Basis of Therapeutics (11thedn), New York: McGraw Hill Companies, Incpp. 629-635.
- Raaman N (2006) Qualitative phytochmeical screening In: Phytochemical techniques. New Delhi: New India publishing agency pp.19-24.
- Gupta S, Duraiswamy B, Satishkumar MN (2013) Peritoneal mast cell stabilization potential of PothosscandensL. Indian J Pharmacol 45: 99-102.
- Norton S (1954) Quantitative determination of mast cell fragmentation by compound 48/80. Br J Pharmacol 9:494-497.
- Bousquet J, Vignola AM, Demoly P (2003) Links between rhinitis and asthma. Allergy 58:691-706.
- Bhende N, Khan I, Shaikh WA, Phadtare JM (1999) Allergy a patient education booklet. National allergy and asthma campaign. India: Hoechst Marion Rousell Ltdpp. 15.
- Lalitharani S, Mohan VR, Maruthupandian A. Pharmacognostical and phytochemical studies on PothosscandensL. Int J Phytomed 2010; 2:277-283.
- Shantha TR, Vasanth Kumar KG, Gopa Kumar K (2007) Pharmacognosy of tala fruits (Borassusflabellier L.). AryaVaidyan20:4:199-205.
- Ram A, Mabalirajan U, Das M, Bhattacharya I, Dinda AK et al. (2006) Glycyrrhizin alleviates experimental allergic asthma in mice. IntImmunopharmacol 6:1468-1477.
- Lacaille-duboisMA,Wagner H (1996) A review of the biological and pharmacological activitiesof saponins.Phytomedicine 2:363-386.
- Mali G R, Dhake SA (2011) A review on herbal antiasthmatics. Orient Pharm Exp Med 11:77-90.
- Tizard IR (1995) Response of mast cell to antigen and chemical mediators of allergic reaction. In: Alexander JL, Rickoff C, Field C, Lubars N, editors. Immunology: An Introduction (4th ed) Florida, Saunders College publishing Harcourt Brace College Publisherspp.456-462.
- Chandrashekhar VM, Halagali KS, Nidavani RB, Shalavadi MH, BiradarBS,et al. (2011)Anti-allergic activity of German chamomile (Matricariarecutita L.) in mast cell mediated allergy model. J Ethnopharmacol 137:336-40.
- Hazekamp A, Verpoorte R, Panthong A (2001) Isolation of bronchodilator flavonoid from the Thia medicinal plant Clerodendrumpetasites. J Ethnopharmacol78:45-49.
- Bellanti JA (1971) Mechanism of tissue injury produced by immunologic reactions. Immunology, Asian ed, Tokyo: W.B. Saunders Co pp. 184.
Open Access Journals
- Aquaculture & Veterinary Science
- Chemistry & Chemical Sciences
- Clinical Sciences
- Engineering
- General Science
- Genetics & Molecular Biology
- Health Care & Nursing
- Immunology & Microbiology
- Materials Science
- Mathematics & Physics
- Medical Sciences
- Neurology & Psychiatry
- Oncology & Cancer Science
- Pharmaceutical Sciences